Mix or match: What do I need to know about vaccine boosters?
FDA and CDC authorized the Moderna and Johnson & Johnson boosters, along with Pfizer’s. Here’s what you need to know now that they’re all available.
Listen 2:48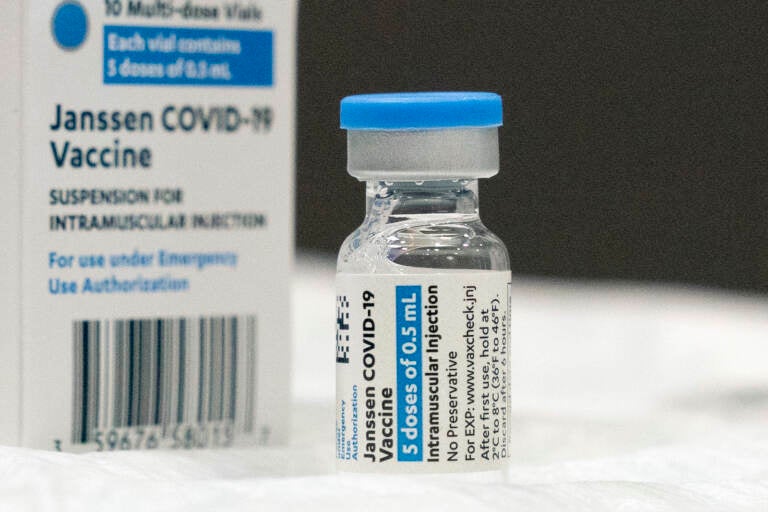
In this March 3, 2021 file photo, a vial of the Johnson & Johnson COVID-19 vaccine is displayed at South Shore University Hospital in Bay Shore, N.Y. U.S. health advisers are meeting Friday, Oct. 15, to tackle who needs boosters of Johnson & Johnson's single-shot COVID-19 vaccine and when. Advisers to the Food and Drug Administration also will examine data suggesting that booster of a competing brand might provide better protection. (AP Photo/Mark Lennihan, File)
Ask us about COVID-19: What questions do you have about the coronavirus and vaccines?
This is one of a series of articles in which reporters from WHYY’s Health Desk Help Desk answer questions about vaccines and COVID-19 submitted by you, our audience.
The Food and Drug Administration and the Centers for Disease Control and Prevention have now authorized COVID-19 boosters for people who received the Moderna and Johnson & Johnson vaccines. (The agencies authorized the Pfizer booster about a month earlier.)
That means boosters are available to a much broader range of individuals, and that the types of vaccines people can get are more diverse.
Who is eligible to receive boosters? And which ones? Here’s what you need to know.
If your first two vaccine doses were Pfizer or Moderna …
You’re eligible to receive a booster at least six months after your second dose if:
- You’re 65 or older.
- You’re over 18 with an underlying health condition.
- You live in a long-term care setting.
- You have a job that puts you at high-risk for catching COVID-19, such as health care worker or teacher.
If you got the Johnson & Johnson shot …
If you’re over 18, you can get a second dose at least two months after your first. This booster is available to more people because the single-dose vaccine has a lower efficacy rate compared to the other two vaccines. Studies show getting a second dose significantly boosts antibody titers, particularly if the second dose is of one of the mRNA vaccines (Pfizer or Moderna).
Which brand of booster should I get?
No matter which type of vaccine you previously received, your booster can be any of the three shots that are now available.
The limited data on mixing vaccines shows that getting a booster different than your original dose or doses provides better protection. The difference is most prominent for people whose original shot was the Johnson & Johnson vaccine.
People already mix vaccine types for other illnesses, including the flu and shingles. Different types of vaccines teach the immune system how to fight viruses in different ways. That means mixing vaccines could make the immune system stronger and smarter, because it will learn how to fight viruses in more than one way, said Dr. Shobha Swaminathan, principal investigator for the Moderna Phase 3 clinical trial at Rutgers New Jersey Medical School.
“When you give the body other types of information, it will be able to produce a more diverse and therefore a more robust immune response,” she said. “When you have a more diverse vaccine strategy, the body’s immune response is more diverse, and therefore will probably be more capable of addressing variants that occur.”
Swaminathan said if you’re eligible for a booster, you should get whatever vaccine type is available.
“If you got Pfizer before and you go to a pharmacy and they only have a Pfizer, I wouldn’t necessarily go looking for Moderna, or vice versa,” she said.
What’s the difference between them?
All three shots of Pfizer are the same, as are both Johnson & Johnson’s doses. However, the Moderna booster is only half the dose of the original, because clinical trials showed it was just as effective as a higher dose. It also means more vaccines will be available to the public.
It’s important to note that a booster is not the same as a third dose.
A booster is meant to top off your immune response.
A third dose is for people who didn’t get an adequate response to the first two doses, such as people who are immunocompromised. People with compromised immune systems who got the Moderna vaccine will get a full dose for their third shot — not the half-dose booster.
What does the future look like?
Many companies are studying boosters that target specific variants, to see if there might be any differences and how frequently people will need them. More research on whether boosters are needed for healthy young adults is needed before health officials can make recommendations for them.

Get daily updates from WHYY News!
WHYY is your source for fact-based, in-depth journalism and information. As a nonprofit organization, we rely on financial support from readers like you. Please give today.





![CoronavirusPandemic_1024x512[1]](https://whyy.org/wp-content/uploads/2020/03/CoronavirusPandemic_1024x5121-300x150.jpg)