Seeking COVID-19 vaccine trial participants from hard-hit communities of color
The coronavirus is disproportionately affecting African Americans and Latinos. Researchers aim to include groups they most want a vaccine to protect.
Listen 1:31
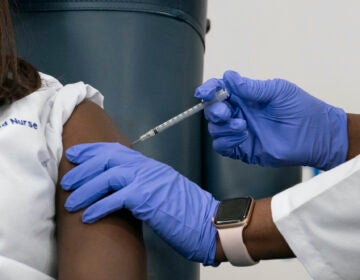
Dr. Chris Pernell is participating in a vaccine trial to fight COVID-19 because her father died of the virus, and her sister is a "long-hauler." (Courtesy of Dr. Chris Pernell)
Are you on the front lines of the coronavirus? Help us report on the pandemic.
Updated 12:45 p.m.
Chris Pernell’s family has been hit hard by the COVID-19 pandemic. She lost her father to the coronavirus. Her sister, a breast cancer survivor, is a “long-hauler” — still fighting various effects of the virus many weeks after having it.
Pernell, 44, also knows that as a Black person, she’s at a higher risk for COVID-19, as two-thirds of people infected are from communities of color. So when the Montclair, New Jersey, resident signed up to volunteer for one of the clinical trials of a vaccine against the virus, it was personal.
“So if I could personally be part of the solution, if I could personally be a part of the prevention science, I wanted to do that,” said Pernell, who also is a public health physician and works as a hospital administrator.
“Knowing how important it is for communities that have been disproportionately impacted, like Black and brown communities, to have access and an opportunity to participate in clinical research, in this vaccine trial in particular, I wanted to be one of those persons who would volunteer,” she said.
“Operation Warp Speed” is what the White House calls the effort to drive vaccine development and provide 300 million doses by January 2021. The New York Times vaccine tracker lists 37 vaccines currently in various stages of human clinical trials.
Researchers want to establish two key things: that the vaccine being tested is effective, and that the vaccine being tested is safe. (On Tuesday, nine vaccine developers, including Moderna, Johnson & Johnson and Pfizer, issued a public pledge not to seek government approval until the shots have proven to be safe and effective.)
Pernell is one of hundreds of people who will participate in a vaccine trial at Rutgers University, which is one of about 100 research sites across the country participating in the biotech company Moderna’s study. Moderna is now in Phase III of the study, which will evaluate whether its mRNA-1273 boosts the immune system to produce enough antibodies against the coronavirus so it does not cause illness in case of infection. There have been 17,458 participants in its nationwide trials so far.
On Aug. 31, Pernell received her first dose of the vaccine after undergoing a lengthy screening process, which included learning about the trial and the vaccine, and undergoing a physical exam, a pregnancy test and blood work. She will return to the clinic in October to get a second and final vaccine. Receiving the trial vaccine felt like getting a flu shot, Pernell said, in that it made her arm sore, but not to the point where she couldn’t perform her regular activities.
She and the other trial participants don’t know if they’ve been given the vaccine or a placebo. They will be observed for an allergic reaction and tested for antibodies, and for a period of about two years, the participants will receive several visits and phone calls.
Pernell said she had no anxiety about participating in the trial. In fact, she said, she was excited, because her father had been a research scientist and professor.
“I felt like I was living out my dad’s purpose in a way by participating in this. So I wasn’t nervous. I was adequately prepped, constantly asked if I have questions,” she said.
Recruiting African Americans into clinical trials historically has been a challenge, however, because of research science’s tainted history of performing risky experiments on them without their consent.
Researchers now aim to inform Black people about how the ethics of clinical trials have changed since those times. They say this communication is particularly important during the pandemic, as communities of color are most affected and therefore are one of the groups scientists want to be sure to protect against the virus.
“I totally understand the skepticism — the Black community has suffered trauma at the hands of academic medicine in the past, it has endured trauma because of medical research in the past that wasn’t done according to the appropriate ethical standards and scientific guidelines,” Pernell said.
“I believe this trial, though it has happened at a faster pace than the public is comfortable with, or any of us are used to, I believe it has not compromised scientific rigor and it has displayed ethics at all times,” she said.
High interest in participating
Academic institutions say the general public’s interest in participating in the vaccine clinical trials has far exceeded anything they’ve seen in the past.
Rutgers, which started screening for its trial last week, received hundreds of applications, phone calls and emails from interested candidates. As of late last week, its study was more than 50% enrolled.
“I think COVID-19 has put us in unprecedented times. I don’t think we’ve ever been in a global pandemic of this urgency that has affected every single person. So, I think that has driven the need for all of us to collectively find a solution to help us get out of this pandemic,” said Dr. Shobha Swaminathan, principal investigator at the Rutgers New Jersey Medical School and medical director of infectious diseases practice at University Hospital.
“We feel blessed, but also know we have a responsibility to our community and our participants to get this right, and make sure the study is conducted and held to the highest ethical standards, and that’s what we strive for, to make sure it’s done right,” she said.

The University of Pennsylvania also has received a great amount of participation interest, said Dr. Emma Meagher, vice dean for clinical research at the Perelman School of Medicine.
In addition to the Moderna trial, Penn also is researching a vaccine created by Inovio Pharmaceuticals, which is using DNA for its trial rather than proteins of the novel coronavirus. The company recently finished Phase I trials and is moving to an accelerated Phase II/III.
Penn has enrolled 95 participants in its Moderna trial so far.
“The minute the Operation Warp Speed was announced by the NIH … academic institutions across the country received floods of offerings to be participants in the trial. It was truly overwhelming how many people wanted to participate,” Meagher said.
“The trials are so much out in the press, they’ve worked their way into being somewhat politicized amongst everything else, so the conversation around vaccines is on everybody’s tongue, because our lives are so affected by COVID,” she said. “There isn’t a member of the living world that isn’t thinking about COVID and the potential for what a vaccine might deliver. So, this is truly unprecedented, and the amount of information people have access to and the types of questions people are asking, this is a very informed public who are truly engaged in what’s happening in vaccine development.”
Her team was not surprised by the interest, she said, but was very relieved that recruitment would not be a challenge.
“There’s incredible pressure to get these trials done. And so, oftentimes the biggest limitation to getting trials done is finding people to participate,” Meagher said. “So, that sense of relief was maybe magnified because of the sense of urgency to get the studies done and get them done well.”
Moderna and researchers at academic institutions say they want a diverse pool of participants to better understand how various age ranges, genders, races and ethnicities respond to the vaccine. The ideal candidate for the trials is someone in a high-risk group, but not so much at risk that it could negatively affect their health. Thus, the goal is for participants to reflect that population most in need of the vaccine.
Those who are at risk of catching the coronavirus because of their work — such as health care workers, first responders, corrections workers, transit workers and those who work in grocery stores — are ideal candidates. Those who are at higher risk of severe complications from COVID-19 also are encouraged to volunteer, such as people over 65 and those with serious health conditions such as high blood pressure and diabetes.
Researchers say it’s important that racial and ethnic groups at higher risk of contracting the virus and having poorer outcomes are included in the trials.
A legacy of mistrust
In the 1800s, Dr. J. Marion Sims performed gynecological surgeries on enslaved Black women without their permission and without anesthesia.
Between 1932 and 1972, the U.S. Public Health Service recruited African American men to participate in the research of syphilis, in what became known as the Tuskegee Experiment. The men were told they were receiving free health care from the U.S. government. The researchers disguised placebos, ineffective methods and diagnostic procedures as treatment, and the men who had syphilis were never informed of their diagnosis, even though it could lead to serious health conditions if left untreated.
Doctors at Johns Hopkins University biopsied Henrietta Lacks’ cervical tumor during her cancer treatment without her knowledge in 1951. Her cells are used to this day to provide medical data. Lacks’ family did not know about the use of her immortalized cell lines until 1975.
Unnecessary hysterectomies, known as Mississippi appendectomies, were performed at teaching hospitals in the South on women of color as practice for medical students.
Today, researchers say they’re making an effort to have conversations with Black people about systemic racism and how clinical trials are performed according to a different ethical standard.
Those conducting COVID-19 vaccine trials are making a concerted effort to recruit Black and Latino volunteers because they have been most affected during the pandemic.
“We want to make sure we provide this opportunity for communities of color such that the clinical trial population is somewhat representative of the U.S. population, so when the results become available we can use that data to say, ‘Yes, this vaccine is safe and effective across all age groups and across all racial and ethnic groups,’” Rutgers’ Swaminathan said.
In addition to choosing academic institutions esteemed for clinical trial and infectious disease research, Moderna also has chosen sites, like the University of Pennsylvania, located in areas with a higher population of Black people and Latinos.
“We have to own up to and acknowledge the broken trust that is an outgrowth of systematic racism,” Pernell said. “As I say, racism has stained the American health care system, and it has impacted Black and brown persons and families. So, we have to own up to that legacy, and we have to work to disrupt and dismantle it.”
She said health care professionals must demonstrate the steps they’re taking to undo those health inequities, and educate the community about clinical research and the risks and benefits.
“There’s two pandemics — there’s this slow pandemic, which is systemic racism that has been with us as long as this nation has been formed, and this fast pandemic, which is this coronavirus,” Pernell said.
“And it’s so important that we as a profession understand the disproportionate burden that racism has placed on the backs of Blacks and browns, Blacks in particular, and an understanding that we can understand the concern, understand the skepticism, and think about culturally fluent ways to have this dialogue in a safe space to say, ‘This is not targeting you unethically,’ ‘This is not anything mandatory.’ Informed consent — what does that mean? And what does that look like? How do we build self-efficacy and collective efficacy?”
Meagher, at Penn, said it’s too soon in the process to evaluate whether Black Americans are reluctant to participate in the vaccine trials. The Penn researchers do not know the racial breakdown of applicants until they are invited to participate. But to date, she said, “We’re doing OK.”
“We’ve got a good representation based on the individuals who have been spoken with about the potential of participation. But it really comes down to, ‘Do they get through the screening period?’ which we’re not there yet, and, ‘Do they stick with their original idea about participating in the trial?’ But it’s looking promising so far,” Meagher said.
To ease concerns about participating in trials, academic institutions like Penn and Rutgers provide a very detailed consent process, outlining what happens during the trial and providing answers to participants’ questions. Researchers also encourage the participation of a next of kin or close friend in the consent process, to ensure that participants have every opportunity to engage. Every study is reviewed by multiple independent boards to ensure participant safety.
Penn is engaged in conversations with community faith leaders to begin to address the distrust felt among many Black people, Meagher said.
“Building community partnerships around research usually are successful when prior participants speak with communities and we facilitate that happening,” Meagher said.
“It takes a lot of work to try and undo all the harm that’s been done. I’m not sure these trials are necessarily going to be our answer, but it is an opportunity for us to start to do some meaningful work and try to address the distrust issue that not only affects the ability to do the trial, but affects our ability to answer the question for the communities whom are most likely to be the recipients if the vaccines get approved,” she said.
Rutgers, too, has a community engagement team for its clinical trials. The university hospital plans community education forums, and in October it will host an event to discuss the disproportionate impact of COVID-19 on communities of color.
“We have definitely made progress, but there’s a lot more work to be done, and I think community education and engagement is an ongoing process, because you always want to keep working with the community, because this has been brewing a long time,” Swaminathan said.
Will a ‘warp speed’ vaccine be safe and effective?
A large proportion of Americans say they will not get a COVID-19 vaccine when one is available.
Thirty-five percent said they would not get a free FDA-approved vaccine if it were ready, according to a Gallup poll. The poll also reported that 4 in 10 non-white Americans, and nearly half of Republicans, indicated they wouldn’t get vaccinated.
“Public trust is waning a little bit,” Meagher said. “In general, people are perplexed, people are confused, they don’t know what end is up. They’re hearing different parts of the same story one day and something different the next day. So, I think in general, the public’s trust in a vaccine that will ultimately be available will entirely depend on the integrity of these trials and the public’s trust they were done properly.
“And I think the fact they are being done in academic medical centers might help that, but equally important is that the FDA makes decisions that are based on the data, and that those decisions are informed by the vaccine being safe and effective. If there’s any question mark around the vaccine being rushed, the vaccine came to market before proven to be effective, ultimately we’re going to pay a price for that hurry-up. Because we will have too many people who will not trust the vaccine, and not want to be vaccinated, and that will defeat what we’re trying to do.”
The Trump administration has asked states to speed up approval for vaccine distribution sites by Nov. 1. And Stephen Hahn, commissioner of the Food and Drug Administration, told the Financial Times that an emergency authorization for a vaccine could be appropriate even before the completion of the third phase of clinical trials.
“I think we all want the vaccine here yesterday,” Swaminathan said with a laugh. “I have elderly parents who can’t wait to get vaccinated, I have young kids who aren’t going to school because of remote learning, so we all are in this together and we all want to get the answer soon.
“I want to emphasize that the scientific process remains intact, and that integrity is critical for the community’s faith and trust — not only in this vaccine, but potentially any other vaccine. So, we are hopeful it will be soon,” she said. “How soon will it be? I don’t know. There are some things you can expedite. You can expedite the enrollment of the study. You can expedite some of the bureaucracy or the review process. But you can’t expedite science. Science will speak for itself.”
Meagher said she agrees that while there is a general societal pressure to get people’s lives as close to normal as possible, scientists want to ensure no corners are being cut.
“I would not say the pressure is to the point of where it could damage the process or the integrity of the trials and how we go about getting the answers,” she said.
Realistically, Meagher said, a vaccine proven safe and effective could be ready in early January 2021. However, she’s concerned the FDA could jump the gun by granting emergency use authorization this year.
Meagher said researchers usually play a significant role in decision-making around when vaccines and treatments are available to the public, and they typically abstain from publishing their research until it’s conclusive.
But the pressures of the pandemic have shifted the goals slightly, because there’s no effective treatment, no vaccine and the health crisis isn’t letting up any time soon.
“So, in that situation, ‘Is something better than nothing?’ begins to operate. [But] ‘Is something better than nothing?’ really shouldn’t be applied to a vaccine,” Meagher said.
“I think the NIH [National Institutes of Health] is quite vocal, and academic organizations are very vocal. But ultimately, it’s the FDA that makes the decision as to what pathway they will proceed, if they want to make a vaccine more broadly available. I think the FDA has articulated they are willing to consider an emergency use authorization, in which efficacy doesn’t have to be demonstrated. I think their willingness to consider that is a departure from normal practice. So, in that situation our voice counts for less. We can articulate, we can try to convene, but ultimately it depends on if the FDA is willing to consider outside opinions in their deliberation process,” Meagher said.
Swaminathan added that while it has been somewhat challenging to launch the trials in a short timeline, it has not been at the cost of the integrity of the trials.
“I think people need to hear that, because I have heard a lot of concerns from people saying, ‘Well it’s going so fast, how can I trust the process?’” she said.
“It’s when everyone puts their mind and will to getting this done. We have diverted the bulk of all the work we were doing to single-handedly get this off the ground because of the global pandemic we are in,” Swaminathan said. “This is a health care crisis that we have not seen in our lifetime, and it’s requiring us to have a coordinated response unlike any before our time.”

Get daily updates from WHYY News!
WHYY is your source for fact-based, in-depth journalism and information. As a nonprofit organization, we rely on financial support from readers like you. Please give today.




![CoronavirusPandemic_1024x512[1]](https://whyy.org/wp-content/uploads/2020/03/CoronavirusPandemic_1024x5121-300x150.jpg)