Kids under 5 could get the COVID-19 vaccine at the end of June. Here’s what you need to know
Kids under 5 may be able to get the COVID-19 vaccine at the end of June. Pediatricians in the region explain what you need to know.
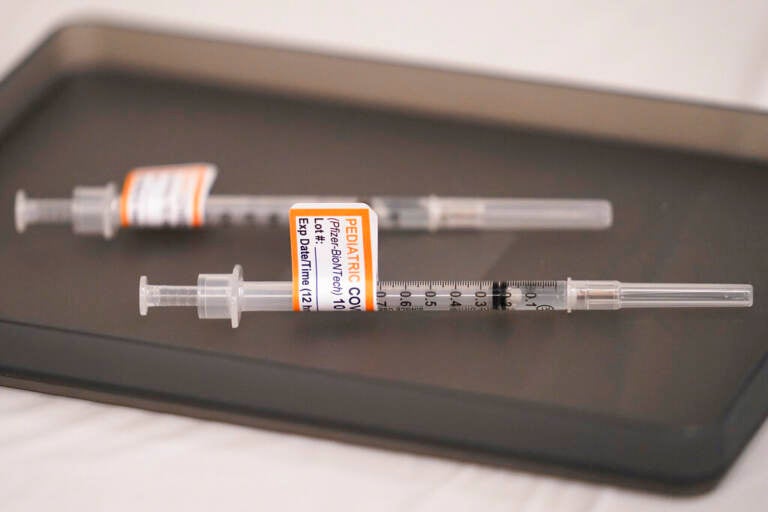
File photo: Prepared Pfizer COVID-19 vaccine syringes for children ages 5 to 11 and adults are displayed on a table at Northwest Community Church in Chicago, Dec. 11, 2021. The Biden administration said Thursday, June 2, 2022, that children under 5 may be able to get their first COVID-19 vaccination doses as soon as June 21, if federal regulators authorize shots for the age group, as expected. (AP Photo/Nam Y. Huh, File)
This is one of a series of articles in which reporters from WHYY’s Health Desk Help Desk answer questions about vaccines and COVID-19 submitted by you, our audience.
The wait for a COVID-19 vaccine has been long for the parents of infants and toddlers.
But they likely won’t have to wait much longer. On Sunday, Food and Drug Administration officials said Pfizer’s pediatric vaccine for kids under 5 appears to be safe and effective. Last week, the agency posted a similar opinion about Moderna’s vaccine for children under 6.
The federal agency’s analysis was submitted ahead of a Wednesday meeting where an advisory committee of experts will vote on whether the shots are ready to be administered. If the shots get a favorable vote, young kids could get their first dose by the end of June.
In February, federal officials postponed approving the Pfizer vaccine for children under 5 after clinical trials showed two doses didn’t produce the desired immune response. This month, the FDA will review a three-dose regimen that Pfizer says boosts antibodies.
Some pediatricians disagreed with the decision to postpone the approval process, arguing that kids under 5 should have received their first two doses while researchers evaluated the third.
“We seem to have had a resurgence in the past couple of months, during the time where we probably could have gotten that vaccine out. So I do think it was a missed opportunity to have made a difference in this wave,” said Dr. Jonathan Miller, a pediatrician and chief of primary care at Nemours Children’s Hospital, Delaware.
“But I am very optimistic about the potential for both Pfizer and Moderna to get approved this month so we can really start the rollout in those younger age groups.”
Though parents have expressed frustration with the hold up on vaccinating kids under 5, others say they’re happy to keep waiting.
A May Kaiser Family Foundation COVID-19 Vaccine Monitor survey found that 38% of parents with kids under 5 plan to wait a while before vaccinating their children, while 27% of them reported they will definitely not get their children vaccinated. Only 18% of parents with children under 5 reported wanting to get their child vaccinated “right away,” and 11% said they’d only do so if required.
Vaccinations among eligible children also have been slower than other age groups. For example, in Philadelphia, just 37% of 5 to 11-year-olds have had at least one dose of the vaccine, compared to a whopping 94% of residents over 12.
Similarly, 35% of 5- to 11-year-olds in Delaware and in New Jersey have had at least one dose.
More than half of parents surveyed in the Vaccine Monitor Survey said they don’t have enough information about the vaccine’s safety and efficacy for children under 5. WHYY’s Health Desk Help Desk reached out to experts in the Delaware Valley to learn more.
Why do kids under 5 need three doses of the mRNA vaccine?
Pfizer’s clinical trial found that two doses did not produce the desired immune response among this age group, meaning their antibody levels weren’t to the level of other age groups.
Dr. Cassie Louis, one of the researchers co-leading the Pfizer vaccine trial for kids at Rutgers University, said she wasn’t surprised by the results, because the dosage is only one-tenth that of Pfizer’s adult COVID-19 vaccine.
However, the results of the third shot are very promising, she said. Clinical trials showed it was 80% effective in preventing symptomatic COVD-19.
“We have seen an increase in immune response, meaning an increase in the body’s first line of defense,” said Louis, an assistant professor of pediatrics at Rutgers. “We’ve seen that the vaccine is helpful for preventing symptomatic COVID, but also for preventing severe infection, meaning requiring hospitalization or having secondary effects like MIS-C [multisystem inflammatory syndrome in children].”
Moderna’s vaccine for kids under 6 is a two-dose regimen, on the other hand. Clinical trials showed that the series was about 40% to 50% effective at preventing milder infections.
The Moderna and Pfizer trials took place at different times of the pandemic, however.
“We have seen an increase in immune response, meaning an increase in the body’s first line of defense,” said Louis, an assistant professor of pediatrics at Rutgers. “We’ve seen that the vaccine is helpful for preventing symptomatic COVID, but also for preventing severe infection, meaning requiring hospitalization or having secondary effects like MIS-C [multisystem inflammatory syndrome in children].”
Should I vaccinate my kid?
Parents have a choice: vaccinate their young kids, or accept their kids’ risk of getting COVID-19, according to Miller, the pediatrician from Nemours.
“We’re at a point where coronavirus has become very, very contagious. Given enough time, it’s likely that your child is going to get COVID,” he said. “And so, you have a decision to make right now. And that decision is, what do you want your child to be exposed to? Do you want them to get the vaccine? Or do you want them to get COVID?”
While we’ve seen by now that COVID infections still occur among the vaccinated, their outcomes are far less harmful, and rarely lethal.
COVID-19, especially among the unvaccinated, can cause serious complications, such as heart and breathing problems. Children also can develop Long COVID, as well as MIS-C – a rare but serious condition associated with COVID-19 that can cause serious gastrointestinal, cardio, and neurological problems. There have been 8,525 MIS-C cases among all children since the beginning of the pandemic. Though much rarer among 5- to 11-year-olds than adults, there have been 364 deaths among this age group.
“We’ve definitely seen some serious long-term side effects of getting COVID,” Miller said. “What we haven’t seen is any serious long-term side effects of getting the COVID vaccine.”
Dr. Vibha Sanwal, a physician at Rainbow Pediatrics in Georgetown, Delaware, said her practice has already ordered the vaccine, and she’s encouraging her patients’ parents to consider the shot for their children.
“They could still get COVID infection in spite of vaccination, but it will be a milder infection. They will not get hospitalized, parents will not lose work, and it’ll be safe for everybody to be vaccinated in the house,” she said.
What if my child already had COVID-19?
Miller and Sanwal say that anyone who has been infected with COVID-19 should get the vaccine, because immunity from both the vaccine and from an infection wanes over time. Some studies also show that getting at least one shot following infection boosts antibodies. Miller said children can get vaccinated 10 days after they’ve recovered from COVID.
What about side effects?
Because the doses are so small, side effects are much milder than they are among adults, Louis said. Common side effects include body aches and headaches. Myocarditis, a type of heart inflammation reported in rare numbers among older kids and young adults, was not reported in the 5 to 11 age group.
“With the trial having gone on for about one year, and having the number of children enrolled that we do, we’ve seen a good time frame for the effect of the vaccine and also for any potential side effects.” Louis said. “And those side effects have remained low throughout the entire trial period.”

Get daily updates from WHYY News!
WHYY is your source for fact-based, in-depth journalism and information. As a nonprofit organization, we rely on financial support from readers like you. Please give today.





![CoronavirusPandemic_1024x512[1]](https://whyy.org/wp-content/uploads/2020/03/CoronavirusPandemic_1024x5121-300x150.jpg)