The failure to report results of clinical trials could hurt you
Listen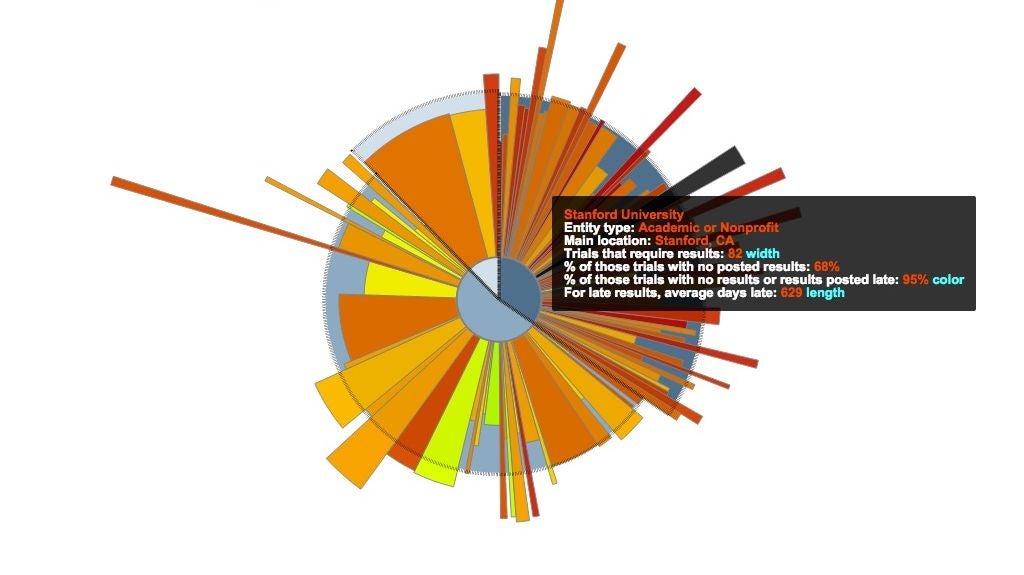
An infographic from the STAT investigation revealing that Stanford University didn't post 68% of their trial results. (Courtesy of STAT)
Big research institutions and pharma companies are failing to report study results, even though they are required to do so, according to a 2008 law. The legislation was intended to make sure that findings from human testing of drugs and medical devices were quickly made public through the website ClinicalTrials.gov.
The failure to report findings could result in unknown negative side effects for patients, plus a hefty fine for the institutions from the FDA. But in yet another plot-twist, the Food and Drug Administration isn’t fining anyone.
We’re joined by reporter Charles Piller. He just published an in-depth investigative report in STAT about this problem, and who it is hurting.
Listen to the full interview above.
WHYY is your source for fact-based, in-depth journalism and information. As a nonprofit organization, we rely on financial support from readers like you. Please give today.



