The Supreme Court fight over an abortion pill: What’s next?
The new abortion controversy comes less than a year after the Supreme Court’s conservative majority overturned Roe v. Wade.
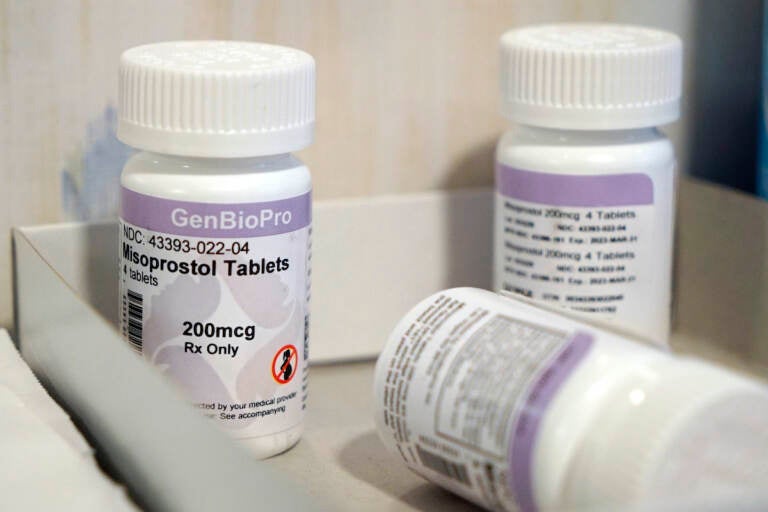
Bottles of the drug misoprostol sit on a table at the West Alabama Women's Center, March 15, 2022, in Tuscaloosa, Ala. (AP Photo/Allen G. Breed, File)
The Supreme Court initially gave itself a deadline of Wednesday to decide whether women seeking access to a widely used abortion pill would face more restrictions while a court case plays out. But on the day of the highly anticipated decision the justices had only this to say: We need more time.
In a one-sentence order, the court said it now expects to act by Friday evening. There was no explanation of the reason for the delay.
The new abortion controversy comes less than a year after the Supreme Court’s conservative majority overturned Roe v. Wade and allowed more than a dozen states to effectively ban abortion outright.
The following is a look at the drug at issue in the new case, how the case got to the nation’s highest court and what the delay might say about what’s going on.
What is mifpristone?
Mifepristone was approved for use by the Food and Drug Administration more than two decades ago. It has been used by more than 5 million women to safely end their pregnancies, and today more than half of women who end a pregnancy rely on the drug, the Justice Department said.
Over the years, the FDA has loosened restrictions on the drug’s use, extending from seven to 10 weeks of pregnancy when it can be used, reducing the dosage needed to safely end a pregnancy, eliminating the requirement to visit a doctor in person to get it and allowing pills to be obtained by mail. The FDA also approved a generic version of mifepristone that its manufacturer, Las Vegas-based GenBioPro, says makes up two-thirds of the domestic market.
Mifepristone is one of two pills used in medication abortions, along with misoprostol. Health care providers have said they could switch to misoprostol only if mifepristone is no longer available or is too hard to obtain. Misoprostol is somewhat less effective in ending pregnancies.
___
How did the case get started?
A lawsuit over mifepristone was filed in Amarillo, Texas, late last year. Alliance Defending Freedom, a conservative Christian legal group, represents the pill’s opponents, who say the FDA’s approval of mifepristone was flawed.
Why Amarillo? U.S. District Judge Matthew Kacsmaryk, who was nominated by then-President Donald Trump, is the sole district court judge there, ensuring that all cases filed in the west Texas city land in front of him. Since taking the bench, he has ruled against President Joe Biden’s administration on several other issues, including immigration and LGBTQ protections.
On April 7, Kacsmaryk issued a ruling that would revoke the FDA’s approval of mifepristone, but he put the decision on hold for a week to allow an appeal.
Complicating matters, however, on the same day Kacsmaryk issued his order, a court in Washington state issued a separate ruling in a lawsuit brought by liberal states seeking to preserve access to mifepristone. The Washington judge, Spokane-based Thomas O. Rice, whom then-President Barack Obama nominated, ordered the FDA not to do anything that might affect the availability of mifepristone in the suing states. The Biden administration has said it is impossible to follow both judges’ directives at the same time.
How did the case go to the Supreme Court?
The Biden administration responded to Kacsmaryk’s ruling by asking the New Orleans-based 5th U.S. Circuit Court of Appeals to prevent it from taking effect for now.
Last week, the appeals court narrowed Kacsmaryk’s ruling so that the initial approval of mifepristone in 2000 is not affected, for now. But it agreed with him that changes the FDA made to relax the rules for prescribing and dispensing the drug should be put on hold. Those rules included expanding when the drug could be taken and allowing for the drug’s delivery through the mail.
The appeals court acted by a 2-1 vote. The judges in the majority, Kurt Engelhardt and Andrew Oldham, are both Trump picks.
The Biden administration and the maker of mifepristone, New York-based Danco Laboratories, appealed to the Supreme Court, saying that allowing the appeals court’s restrictions to take effect would cause chaos. Facing a tight deadline, the Supreme Court gave itself some breathing room and issued an order suggesting it would act by Wednesday evening. That timeline was extended to Friday, the day the justices will hold a previously scheduled private conference.
The justices could talk about the issue further then. The additional time could also be part of an effort to craft an order that has broad support among the nine justices. Or one or more justices might be writing a separate opinion and asked for a couple of extra days.
___
What could happen next?
The Supreme Court’s delay suggests a maddening reality about an institution that ordinarily adheres to a schedule that hasn’t changed much in years: Even experts can be in the dark about when the court will decide things and how.
Cases are argued over seven months from October to April, and the most important decisions typically come right before the justices take a long summer break. The court does not say which cases it plans to hand down on a given day, and the court, in a search for consensus, will sometimes pass on the biggest issues it faces and decide a very small legal point.
But nowhere is the uncertainty as great as a separate category of cases that have come to be known as the shadow docket.
Apart from death row inmates seeking 11th-hour reprieves, shadow docket cases generally involve emergency appeals to the justices before lower courts have reached final decisions. That includes the mifepristone case.
When the justices consider this set of cases, they don’t usually have a deadline to act. A few years back, an order concerning an elections case in Texas came in the wee hours of a Saturday morning for no reason other than that’s when Justice Ruth Bader Ginsburg finished work on her dissenting opinion.
WHYY is your source for fact-based, in-depth journalism and information. As a nonprofit organization, we rely on financial support from readers like you. Please give today.