Parenting of the future: Many embryos, each with DNA profile
You've probably read about concerns over "designer babies," whose DNA is shaped by gene editing.
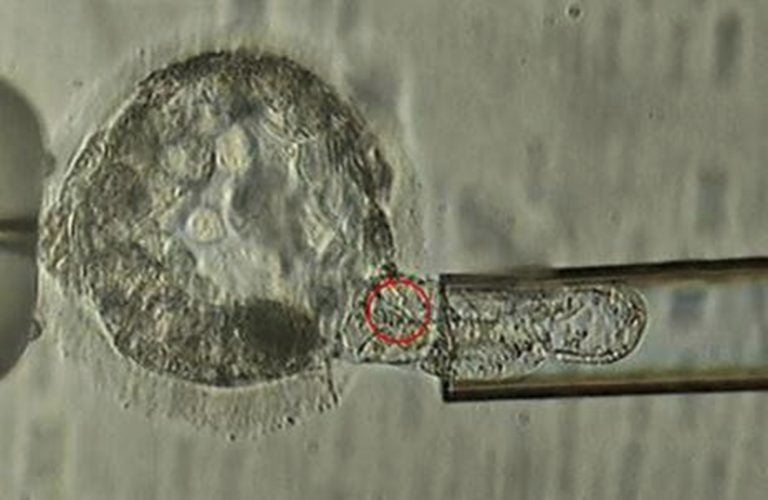
This undated microscope image provided by the American Society for Reproductive Medicine in January 2018 shows a trophectoderm biopsy, in which cells from the outer layer of an embryo that develop into the placenta and amniotic membranes are removed and can be used for genetic testing. When a couple is known to be at risk for having a child with a specific genetic disorder, the woman undergoes a procedure to remove some of her eggs. After fertilization, some cells can be plucked from the embryos and examined to identify those without carry the disease-causing abnormality. (ASRM via AP)
So you want to have a baby.
Would you like a dark-haired girl with a high risk of someday getting colon cancer, but a good chance of above-average music ability?
Or would you prefer a girl with a good prospect for high SAT scores and a good shot at being athletic, but who also is likely to run an above-average risk of bipolar disorder and lupus as an adult?
How about a boy with a good shot at having musical ability and dodging asthma, but who also would be predisposed to cataracts and type 2 diabetes?
Confused? You’re just getting started. There are dozens more choices for which of your embryos should be placed in the womb to become your child.
That’s the future a biomedical ethics expert envisions for 20 to 40 years from now — soon enough that today’s children may face it when they start their own families.
“The majority of babies of people who have good health coverage will be conceived this way,” predicts Henry Greely, a Stanford University law professor who works in bioethics.
You’ve probably read about concerns over “designer babies,” whose DNA is shaped by gene editing. Greely is focused on a different technology that has gotten much less attention: In a startling bit of biological alchemy, scientists have shown that in mice, they can turn ordinary cells into sperm and eggs.
It’s too soon to know if it could be done in people. But if it can, it could become a powerful infertility treatment, permitting genetic parenthood for people who can’t make their own sperm or eggs.
It also would mean that a woman who wants to get pregnant could produce dozens more eggs per attempt than with the current procedure of harvesting some from her ovaries.
And that means a lot of choices.
An array of embryos
Here’s what Greely envisions: A man and woman walk into a fertility clinic. The man drops off some sperm. The woman leaves some skin cells, which are turned into eggs and fertilized with the man’s sperm.
Unlike in vitro fertilization today, which typically yields around eight eggs per try, the new method could result in 100 embryos.
The embryos’ complete library of DNA would be decoded and analyzed to reveal genetic predispositions, both for disease and personal traits. The man and woman would get dossiers on the embryos that pass minimum tests for suitability.
Out of, say, 80 suitable embryos, the couple would then choose one or two to implant.
The possibilities don’t stop there. The technology might also help open the door to same-sex couples having children genetically related to both of them, though the additional twist of making eggs from men or sperm from women would be a huge biological challenge.
More worrisome is the so-called Brad Pitt scenario: We all shed a bit of sloughed-off DNA every day, like on the lip of a coffee cup. Such discarded material could be secretly snatched up to turn an unwitting celebrity into a genetic parent.
It is a long way in the future, but real life is already creeping toward it. Some scientists are trying to make human eggs and sperm in the lab. They are working with “iPS cells,” which are ordinary body cells that have been morphed into a malleable state.
Amander Clark of the University of California, Los Angeles, says her goal is to aid basic research into why some people are infertile. She acknowledges the technique might itself be used to treat some infertility, particularly in young people made sterile by cancer treatments.
As for decoding the complete DNA library of embryos, Dr. Louanne Hudgins, who studies prenatal genetic screening and diagnosis at Stanford, says some pregnant patients there say they’ve already had fertility clinics do that. They didn’t reveal why, Hudgins said.
Hudgins, who’s president of the American College of Medical Genetics and Genomics, said no national medical association has endorsed decoding all the DNA of an embryo, which is called its genome. So she believes no insurance company would pay for that now.
‘Easy’ prenatal diagnosis
Greely, who lays out his ideas in a book called “The End of Sex and the Future of Human Reproduction,” calls his vision “easy PGD,” or prenatal genetic diagnosis.
Ordinary PGD has been done for decades. When a couple is known to be at risk for having a child with a specific genetic disorder, such as cystic fibrosis or sickle cell anemia, the woman undergoes a procedure to remove some eggs. After fertilization, some cells are plucked from the embryos and examined to identify those without carry the disease-causing abnormality.
That procedure looks for a specific problem in a few embryos, not entire genomes from dozens of them. If a couple wants to select a “super baby,” says Dr. Richard Scott Jr., a founding partner of Reproductive Medicine Associates of New Jersey, “we tell them we can’t do it.”
In fact, Scott and others say, even wide-ranging analysis would not provide a precise forecast of how a child will turn out.
If DNA is the hardware, there’s also the software: chemical modifications that determine when and where particular genes turn on and off. Much of this “epigenome” would develop after an embryo’s genes are sampled, Scott said.
“Your child may not turn out to be the three-sport All-American at Stanford,” because “the epigenome didn’t work out,” Scott said.
Greely agrees that predictions about behavioral traits like intelligence and athletic ability will be imprecise, because of epigenetics and because of basic uncertainties about what genes are involved and how they interact. And a person’s upbringing and life experiences have a big effect.
What would couples do?
Even if the predictions aren’t perfect, would couples want to take steps to control their child’s genetics? Many experts doubt it.
Only a “very small minority” seek a perfect baby, says Stanford’s Hudgins. In her practice, she said she often finds women pass up all screening because they figure the baby’s fate is “in God’s hands.”
Dr. James Grifo of the New York University Fertility Center also questions how popular the idea would be.
“No patient has ever came to me and said, ‘I want a designer baby,'” said Grifo, who’s performed in vitro fertilization since 1988.
Greely doubts that influencing brainpower or athleticism would be a major draw for parents. Instead, he thinks they would care most about avoiding awful diseases that strike in infancy or childhood. They’ll probably be less concerned about illnesses that might show up later in life, such as Alzheimer’s or Parkinson’s. For one thing, he says, parents-to-be may see them as becoming treatable by the time a child becomes vulnerable.
He thinks easy PGD is coming, and it would be better if properly handled. He says it should be proven safe, subsidized, monitored for long-term effects, and regulated so that parents can choose whether to use it and decide what embryonic traits to focus on. And he’d outlaw stealing somebody’s DNA and unwittingly making them a parent.
Others see pitfalls
Once the genetic profile is done, could it come back to haunt a child if, say, a life insurer or nursing home demanded to see it to assess disease risk? How would the large number of rejected embryos be handled ethically and politically?
Perhaps future regulation could limit the number of embryos created, as well as what traits a couple could select for, said I. Glenn Cohen, a Harvard law professor.
Lori B. Andrews, a professor at the Chicago-Kent College of Law, summed up her views in a review of Greely’s book.
“The idea of easy PGD,” she wrote, “should make us uneasy indeed.”
Still, even some who doubt the idea’s feasibility say Greely is right to raise the issue.
“It’s certainly something we have to take seriously and think through now,” said Marcy Darnovsky, who writes on the politics of human biotechnology as executive director of the Center for Genetics and Society in Berkeley, California. “This is not just a technical or science question.”
Follow Malcolm Ritter at @MalcolmRitter . His recent work can be found here .
This Associated Press series was produced in partnership with the Howard Hughes Medical Institute’s Department of Science Education. The AP is solely responsible for all content.
WHYY is your source for fact-based, in-depth journalism and information. As a nonprofit organization, we rely on financial support from readers like you. Please give today.




