CHOP researchers make progress on sickle cell disease treatment
Listen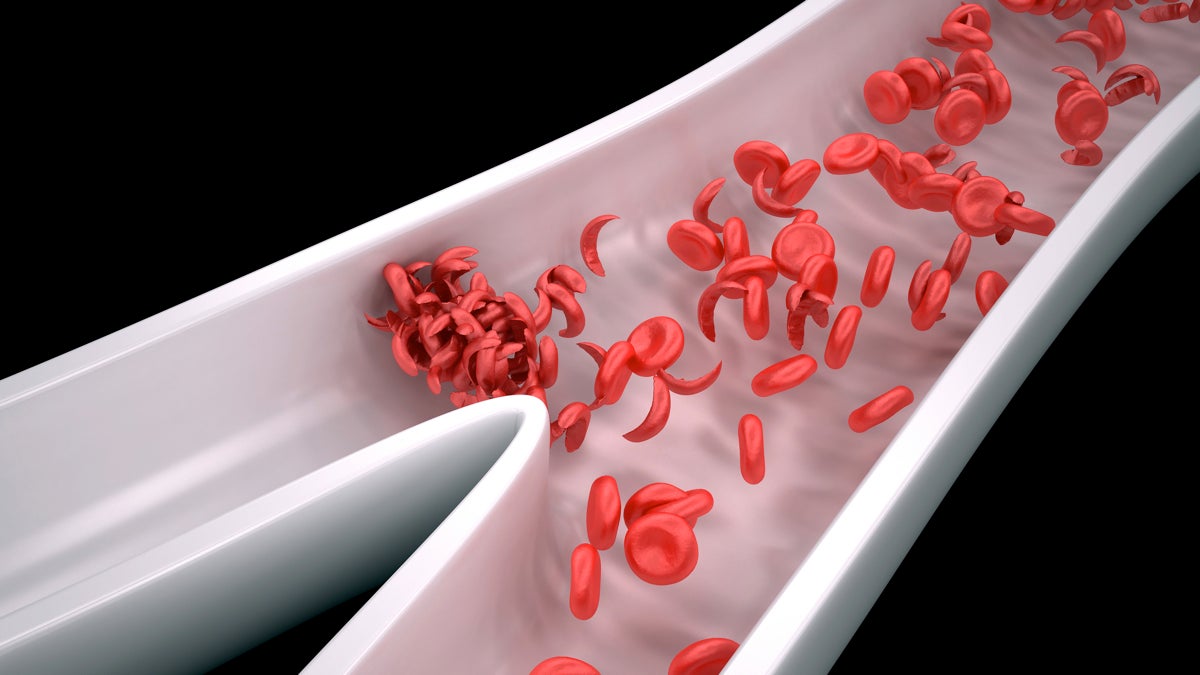
Sickle cell disease results from a mutation in hemoglobin — the protein that carries oxygen in the blood — that causes red blood cells to be crescent-shaped.(Shutterstock)
There’s only one way to cure sickle cell disease: a bone marrow transplant. But it’s difficult to find donors, and the treatment carries risks. Now, researchers at the Children’s Hospital of Philadelphia are having early success with an alternative approach to treatment.
Most common in those of African descent, sickle cell disease results from a mutation in hemoglobin — the protein that carries oxygen in the blood — that causes red blood cells to be crescent-shaped. That leaves the cells less flexible and stickier; they clump together and can block blood vessels, damaging organs.
“The symptoms include severe pain, anemia, chest pain and breathing problems, stroke, infections, and many others,” said CHOP scientist Gerd Blobel. “So it’s a really serious disease.”
But Blobel notes that only adult hemoglobin is mutated. Prior to birth, sickle cell babies use fetal hemoglobin, which isn’t mutated, and they are protected.
“Many years ago [scientists] discovered that patients that failed to completely turn off this fetal hemoglobin after birth, and maintain fetal globin expression throughout adulthood, they tend to experience a much milder course of the disease,” he said.
Ever since, researchers have been trying to come up with ways to increase fetal hemoglobin. In fact, the only FDA-approved drug to treat sickle cell disease, hydroxyurea, works by doing so — but not all patients always take it.
Blobel’s approach draws on years of basic research investigating how DNA physically interacts with itself and other proteins to activate genes in cells. Key to the process are loops of DNA he likens to shoelaces: the knot is a hub for activity, leading to a gene being turned on.
“We tried to figure out how are these loops formed, what factors control these, and then, in the process, we then found a way to actually alter these loop structures of chromosomes specifically at the genes that make hemoglobin,” said Blobel.
The team introduced specialized proteins into cells to force a new looping paradigm, essentially relocating the knot closer to the fetal gene and farther away from the adult gene.
For human cells growing in culture, they succeeded in switching 85 percent of total hemoglobin to the fetal kind. The results were published this month in the journal Cell.
“We’re not only turning on the fetal hemoglobin gene, but we’re also turning off the toxic, bad form, of hemoglobin that causes sickle cell disease,” said Blobel.
That differs from other experimental gene therapy approaches that increase the amount of fetal hemoglobin, but leave plenty of adult protein around that can compete with the fetal form.
Blobel says the next goal is curing the disease in mice. If successful, that could lead to clinical trials in two to three years.
WHYY is your source for fact-based, in-depth journalism and information. As a nonprofit organization, we rely on financial support from readers like you. Please give today.

