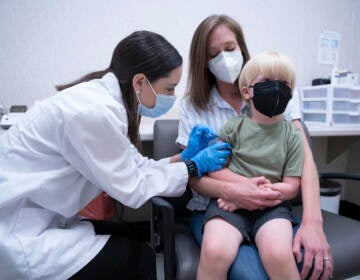
CDC advisory panel recommends coronavirus vaccines for adolescents aged 12-15
A Pfizer vaccine vial and syringe. A CDC advisory panel has recommended the Pfizer/BioNTech vaccine be administered to adolescents age 12 to 15. (Jaap Arriens/NurPhoto via Getty Images)
An independent federal advisory committee on Wednesday recommended that the Pfizer-BioNTech coronavirus vaccine be administered to adolescents aged 12 to 15 — a move expected to be quickly approved by the Centers for Disease Control and Prevention.
Wednesday’s decision could open up a new front in the fight against COVID-19 after the Food and Drug Administration’s extension of its emergency use authorization for the Pfizer vaccine to cover adolescents. The vote was 14 in favor with one recusal.
Previously, coronavirus vaccines in the U.S. had only been approved for people age 16 and older. Pfizer is the first vaccine manufacturer to gain emergency use authorization for younger Americans after it demonstrated in a March clinical trial that its vaccine was 100% effective in preventing COVID-19 in study participants who were aged 12 to 15.
During a meeting lasting nearly four hours, the committee heard details of studies showing the vaccine prevented COVID-19 in more than 1,000 adolescents in the age range, while 16 cases occurred in those who got a placebo. There were no serious side effects reported.
Public health officials, including those from the CDC and the independent advisors on the committee, said the vaccine will further help control the pandemic in the U.S. and in other countries that typically follow the U.S. lead. Data presented by the CDC showed that about 20% of COVID-19 cases in the U.S. have been in children and adolescents 17 years of age and younger.
The American Academy of Pediatrics endorsed the move in a statement read during the meeting.
“This is truly an exciting development that allows us to protect a large population of children and help them regain their lives after a really rough year,” said AAP President Lee Savio Beers. “As a pediatrician and a parent, I have looked forward to getting my own children and patients vaccinated, and I am thrilled that those ages 12 and older can now be protected. The data continue to show that this vaccine is safe and effective. I urge all parents to call their pediatrician to learn more about how to get their children and teens vaccinated.”
Comments from the public during an open session included questions of whether the relatively low risk of serious COVID-19 complications in children justifies the use of the vaccine in this age group before more studies are performed. The study included more than 2,000 adolescents, but was criticized by some public commenters for not being of sufficient size. Commenters also expressed concern that the studies were not sufficient to demonstrate whether there are any long-term effects associated with this or other COVID-19 vaccines.
CDC and company officials stressed study findings that show that the vaccine is extremely safe in this age group, paralleling what has been seen in adults for several months now. More than 150 million people have received at least one dose of one of the vaccines with a very low rate of serious side effects.
The authorization and endorsement come in time for many adolescents to get vaccinated before participating in summer activities such as camping and group sports. Efforts to get as many adolescents vaccinated as possible before school restarts in August and September are already underway.
In the pediatric studies, the CDC and FDA evaluated the same two-dose regimen separated by three weeks that is used for people aged 16 and up.
While children and adolescents generally have milder symptoms if they contract COVID-19 compared to adults, they can nonetheless pass the disease on to others. That has parents worried about the prospect of schools fully re-opening in the fall.
“I think we should be in full school, full in-person school, in the fall,” CDC Director Dr. Rochelle Walensky said at a CNBC health summit on Tuesday.
Despite the Biden administration’s push to get enough Americans inoculated against COVID-19 to establish “herd immunity,” the pace of vaccinations in the U.S. has slowed in recent weeks, with many adults opting not to get the shots.
CDC data show that just over a third of Americans are fully vaccinated, with 46% having received at least one dose. While surveys have showed that only about 46% to 60% of parents said they planned to have their adolescent children immunized, opening up vaccinations for millions of adolescents would likely get the U.S. closer to the goal of herd immunity.
In March, both Pfizer and Moderna launched pediatric trials of their vaccines in children as young as 6 months old. Results are expected in the early fall. Neither company has applied to the FDA for emergency use authorization in children younger than 12.
Johnson & Johnson resumed shipments of its one-dose vaccine last month after U.S. federal regulators ended a 10-day pause in administering the doses while a potential link to a rare form of blood clot was examined.
The Moderna and Johnson & Johnson vaccines are currently only approved for for adults aged 18 and older.
Meanwhile, states had already begun sending the Pfizer vaccine to pediatricians in anticipation of Wednesday’s recommendation, according to The Associated Press.
9(MDAzMzI1ODY3MDEyMzkzOTE3NjIxNDg3MQ001))


![CoronavirusPandemic_1024x512[1]](https://whyy.org/wp-content/uploads/2020/03/CoronavirusPandemic_1024x5121-300x150.jpg)